

・We organized the 11th web meeting of the International Leading Research Project (5.12, 2026)
・We organized the tenth web meeting of the International Leading Research Project (4.16, 2026)
・Congratulations to Dr. Shunsuke Tanigawa on his move to Yamaguchi University as a professor! (4.1, 2026) Prof. Nishinakamura’s Congratulatory Remarks
・Kazuki Honda joined our lab. Welcome! (4.1, 2026)
・Momoha Sugasaki graduated from the master’s program (3.25, 2026)
・Fourteen high school students from Ikeda Gakuen in Kagoshima visited our institute (3.18, 2026)
・We organized the ninth web meeting of the International Leading Research Project (3.10, 2026)
・We organized the eighth web meeting of the International Leading Research Project (2.10, 2026)
・Prof. Nishinakamura gave a presentation at the Takeda Science Foundation Symposium, “Organoid 4D.” (1.23, 2026)
・We organized the seventh web meeting of the International Leading Research Project (1.13, 2026)
・Prof. Nishinakamura gave a lecture at Asia Pacific Congress of Nephrology in Taiwan (12.6, 2025)
・Prof. Nishinakamura gave a lecture at the Chinese Academy of Sciences in Shanghai (11.26, 2025)
・Prof. Nishinakamura gave a lecture at the Nature Conference in Hangzhou, China (11.19, 2025)
・Dr. Fahim Haque, an associate professor at BRAC University in Bangladesh, visited our lab to collaborate (11.13-29, 2025)
・We organized the sixth web meeting of the International Leading Research Project (11.11, 2025)
・Dr. Shunsuke Tanigawa gave a lecture at the 6th Food for Health International Conference & Life Science Symposium in China (10.15, 2025)
・We organized the fifth web meeting of the International Leading Research Project (10.14, 2025)
・We invited Prof. Hiroshi Kimura from Tokai University to give a lecture (10.7, 2025)
・Dr. Shunsuke Tanigawa received a JST Sakigake grant (9.18, 2025)
・Qipeng Sun joined the lab as a graduate student. Welcome! (10.1, 2025)
・Ziyu He obtained his Ph.D. and became a postdoctoral researcher. Congratulations! (10.1, 2025)
・Keishiro Furuie and Ola Shalaby presented posters at the IMEG International Symposium (9.30, 2025)
・Ola Shalaby obtained her Ph.D. and graduated from the lab. Congratulations! (9.25, 2025)
・Prof. Nishinakamura gave a lecture at the ASHBi symposium in Kyoto (9.20, 2025)
・Diasuke Inoue and Yutaro Ibi gave oral presentations at the Developmental Nephrology meeting (9.13, 2025)
・We organized the fourth web meeting of the International Leading Research Project (9.9, 2025)
・Prof. Nishinakamura gave a lecture at the Japan Human Cell Society meeting in Tokyo (9.6, 2025)
・The figure from Ola Shalaby’s paper is featured on the cover of the American Journal of Pathology (9.4, 2025)
・Dr. Shunsuke Tanigawa taught an intensive lecture course at Kagoshima University (9.3-5, 2025)
・Dr. Shunsuke Tanigawa taught a special lecture at Shinwa High School (8.26, 2025)
・Dr. Shunsuke Tanigawa gave a lecture at Kuroshio Conference in Ehime (8.23, 2025)
・International Leading Research “Creating a kidney”: Kumamoto Retreat and Joint seminars (7.25-28, 2025)
・Dr. Tanigawa gave an oral presentation at the annual meeting of the Japanese Society of Developmental Biologists (7.16, 2025)
・Yutaro Ibi’s paper on ureteral organoids was published in Nature Communications [NewPress] [Publications] (6.20, 2025)
・Ola Shalaby’s paper was published in the American Journal of Pathology [NewPress] [Publications](6.9, 2025)
・Dr. Yuhao Wang was awarded with the Sakagawa Medical Scholarship (4.16, 2025)
・Nine years since the Kumamoto Earthquakes (4.14, 2025)
・Daisuke Inoue presented his data at the first web meeting of the International Leading Research Project (4.8, 2025)
・Yuhao Wang, Ziyu He, and Momoko Hattori joined the lab (4.1, 2025)
・Drs. Ibi and Ide graduated from the lab. We wish you both the best of luck in your future endeavors (3.31, 2025)
・Prof. Nishinakamura gave a seminar at the Alport Workshop (3.21, 2025)
・We have launched the International Leading Research “Creating a kidney” (12.3, 2024)
・Hiroshi Ide’s paper was published in Scientific Reports (12.5, 2024)
・Dr. Tanigawa gave an oral presentation at the International Symposium of the Institute Network for Biomedical Sciences (10.5, 2024)
・Dr. Tanigawa gave a lecture at the Western Regional Meeting of the Japanese Society of Nephrology (10.5, 2024)
・Dr. Young-Hyun Go joined our lab as a postdoctoral researcher (9.2, 2024)
・Prof. Nishinakamura gave a lecture at Korean Society of Stem Cell Research (8.29, 2024)
・Prof. Nishinakamura gave a lecture at Gordon Research Conference (8.14, 2024)
・Asmin Mizrak from Turkey spent 4 weeks in the lab as a student intern (7.2, 2024)
・Prof. Nishinakamura gave a lecture at the Japanese Society of Regenerative Medicine (3.22, 2024)
・Three researchers from Lithuania and China stayed in the lab for two weeks (3.1, 2024)
・Prof. Nishinakamura has received the Academic Award of the Japan Kidney Foundation (2.22, 2024)
・Mandakh Ganbaatar from Australia spent 6 weeks in the lab as a student intern (2.22, 2024)
・Prof. Nishinakamura gave a lecture at Sun Yat-sen University in China (12.12, 2023)
・Dr. Tanigawa gave an oral presentation at the annual meeting of the Molecular Biology Society of Japan (12.6, 2023)
・Prof. Nishinakamura gave a lecture to the patients in Okinawa (11.20, 2023)
・Prof. Nishinakamura gave a lecture at Lithuanian University of Health Sciences (10.19, 2023)
・Prof. Nishinakamura gave a lecture at the International Workshop on Developmental Nephrology in Canada (8.16, 2023)
・The review article by Prof. Nishinakamura has been published in Cell Stem Cell (8.4. 2023)
・Dr. Etsuko Tanaka (currently Assistant Professor, Miyazaki University) received the Morita Prize from the Japanese Society of Pediatric Nephrology (7.1, 2023). Congratulations!
・Prof. Nishinakamura gave a talk at Cold Spring Harbor Laboratory in New York (6.1, 2023)
・Prof. Nishinakamura gave a lecture at the Japanese Society of Regenerative Medicine (3.23, 2023)
・Dr. Shohei Kuraoka left the lab to join Harvard University. We wish you further success (3.27, 2023)
・An article based on an interview with Prof. Nishinakamura was published in Nature (3.10, 2023)
・Prof. Nishinakamura gave a lecture at Cold Spring Harbor Asia (3.9, 2023)
・Dr. Shunsuke Tanigawa received a grant from the FOREST program (1.27, 2023)
・Prof. Nishinakamura will appear on the NHK BS program “Humanience” (1. 17, 2023)
・Prof. Nishinakamura gave a special lecture at the Japanese Society of Nephrology (11.18, 2022)
・Prof. Nishinakamura gave a lecture at the American Society of Nephrology in Florida (11.3, 2022)
・Prof. Nishinakamura gave a lecture in Science and Me series (7.1, 2022)
・Tanigawa made an oral presentation at ISSCR in San Francisco (6.17, 2022)
・The paper by Dr. Tanigawa is featured in a Nature Portfolio collection (6.10, 2022)
・Dr. Etsuko Tanaka won the best presentation award from the Japanese Society for Pediatric Nephrology (5.22, 2022)
・Prof. Nishinakamura steps down as Deputy Director (3.31, 2022)
・Shunsuke Tanigawa won the Best Presentation Award at the 6th Podocyte Conference in Japan (3.26, 2022)
・Our organoid technology was licensed to Trestle Biotherapeutics (2.15, 2022)
・The paper by Shunsuke Tanigawa and Etsuko Tanaka was published in Nature Communications [NewPress] [Publications] (2.1, 2022)
・Shankhajit De left the lab. We wish you further success in Hiroshima (11.30, 2021)
・Prof. Nishinakamura gave a seminar in GfE School (11.28, 2021)
・Prof. Nishinakamura gave a seminar in ASHBi International Symposium (11.9, 2021)
・Prof. Nishinakamura gave a seminar in the American Society of Nephrology meeting (11.4, 2021)
・Tomoko Ohmori’s paper was published in Scientific Reports [Publications] (2.17, 2021)
・Hidekazu Naganuma’s paper was published in Developmental Biology [NewPress] [Publications] (11.14, 2020)
・Prof. Nishinakamura gave a seminar in the American Society of Nephrology meeting (10.22, 2020)
・Prof. Nishinakamura gave a seminar in the ISSCR webinar (10.8, 2020)
・Shohei Kuraoka’s paper was published in J Am Soc Nephrol [NewPress] [Publications] (8.3, 2020)
・Prof. Nishinakamura gave a seminar at the University of Pennsylvania (12.16, 2019)
・Mazharul Islam left the lab. We wish you success in Sweden (2019.10.31)
・Yasuhiro Yoshimura’s review was published in Kidney International [Publications](10.21, 2019)
・Our review was published in Nature Reviews Nephrology [Publications](8.5, 2019)
・Shunsuke Tanigawa’s paper was published in Stem Cell Reports [NewPress] [Publications](8.2, 2019)
・Prof. Nishinakamura gave a lecture in the plenary session of ISSCR (6.27, 2019)
・Shunsuke Tanigawa made an oral presentation at the annual meeting of the Japanese Society of Nephrology (6.23, 2019)
・Yasuhiro Yoshimura won the Presidential Award at the annual meeting of the Japanese Society of Nephrology (6.22, 2019)
・Prof. Nishinakamura gave a seminar in India (3.6, 2019)
・Yoichi Murakami’s paper was published in Scientific Reports [Publications] (2.4, 2019)
・Yasuhiro Yoshimura’s paper was published in the Journal of the American Society of Nephrology [NewPress] [Publications] (1.11, 2019)
・Dr. Akio Koyabashi joined our lab (1.1, 2019)
・Prof. Nishinakamura organized a symposium at the annual meeting of the Molecular Biology Society of Japan (11.29, 2018)
・Prof. Nishinakamura gave two lectures at the American Society of Nephrology meeting in San Diego (10.23&25, 2018)
・Shunsuke Tanigawa obtained the best poster award in the International Symposium of the Institute Network for Biomedical Sciences (10.19, 2018)
・Shunsuke Tanigawa’s paper was published in Stem Cell Reports [NewPress] [Publications] (8.30, 2018)
・Shunsuke Tanigawa gave a seminar at the annual meeting of Japanese Society of Nephrology (6.10, 2018)
・Yasuhiro Yoshimura received Poster Award at the International Podocyte Conference in Montreal (6.2, 2018)
・Prof. Nishinakamura gave a seminar at the International Podocyte Conference in Montreal (5.31, 2018)
・Prof. Nishinakamura gave a seminar in South Korea (5.17, 2018)
・Prof. Nishinakamura gave a seminar in China (4.14, 2018)
・Atsuhiro Taguchi’s paper was published in Cell Stem Cell [NewPress] [Publications] (11.10, 2017)
・Dr. Shankhajit De joined the lab as a postdoctoral fellow (10.2, 2017)
・Fahim Haque obtained the PhD degree and graduated from the lab. We wish you further success in the future. (9. 25, 2017)
・Atsuhiro Taguchi left the lab. We wish you further success in Germany (9. 1, 2017)
・Yusuke Kaku’s paper was published in Scientific Reports [NewPress] [Publications] (7.3, 2017)
・The meeting review by Prof. Nishinakamura was published in Development [Publications] (6.20, 2017)
・Fahim Haque’s paper was published in Developmental Biology [NewPress][Publications](6.10, 2017)
・A message from the Director regarding the earthquakes (4.14, 2017)
・Nishinakamura gives a seminar at RIKEN CDB symposium (3.28, 2017)
・Taguchi gives a seminar at Germany-Japan joint meeting in Germany (3.16, 2017)
・The review by Dr. Tanigawa was published in Kidney International (10. 17, 2016)
・A message from the Director regarding the earthquakes (10. 14, 2016)
・Nishinakamura gave a seminar in the EMBO/EMBL symposium in Germany (10.14, 2016)
・Ryuichi Nishinakamura gives a seminar at Santa Cruz Development Biology Meeting (8.15, 2016)
・A message from the Director regarding the earthquakes (7.14, 2016)
・Messages from the Director regarding the earthquakes(5.2,2016)
・Shunsuke Tanigawa’s paper is published in Cell Reports.[NewPress] [Publications] [Facebook](4.15, 2016)
・Ryuichi Nishinakamura is appointed Director of the Institute. (4.1, 2016)
・Ryuichi Nishinakamura gives a seminar at the CiRA/ISSCR joint symposium. (3.24, 2016)
・Ryuichi Nishinakamura’s review is published in Nat Rev Nephrol. (2.1, 2016)
・Sazia Sharmin’s paper is published in J Am Soc Nephrol. Sazia has obtained a PhD degree. (11.20, 2015)
・Tomoko Ohmori’s paper is published in Scientific Reports. (10.29, 2015)
・Shunsuke Tanigawa’s paper is published in Stem Cell Reports. (8.27, 2015)
Generation of 3D kidney from pluripotent stem cells
By focusing on an often-overlooked tissue type of organoid generation research, a type of organ tissue made up of various support and connective tissues called the stroma, our team led by Prof. Ryuichi Nishinakamura (Department of Kidney Development, Institute of Molecular Embryology and Genetics (IMEG), Kumamoto University) were able to generate the last of a three-part puzzle that we had been working on for several years. Once the three pieces were combined, the resulting structure was found to be kidney-like in its architecture. We believe that our work will be used to advance kidney research and even lead to a transplantable organ in the future.
The kidney is a very important organ for continued good health because it acts as a filter to extract waste and excess water from blood. It is a complex organ that develops from the combination of three components. Protocols have already been established by various research teams, including ours, to induce two of the components (the nephron progenitor and the ureteric bud) from mouse ES cells.
In this, our most recent work, Shunsuke Tanigawa and Etsuko Tanaka in the team have developed a method to induce the third and final component, kidney-specific stromal progenitor, in mice. Furthermore, by combining these three components in vitro, we were able to generate a kidney-like 3D tissue, consisting of extensively branched ureteric buds and several other kidney-specific structures, such as glomeruli and renal tubules (Figure).
We believe that this is the first ever report on the in-lab generation of such a complex kidney structure from scratch. Our team has already succeeded in inducing the first two components from human iPS cells (Cell Stem Cell 2014, Cell Stem Cell 2017). If this last component can also be generated from human cells, a similarly complex human kidney should be achievable. We hope to use our developments to screen drugs for various diseases, and for transplantation in the long run.
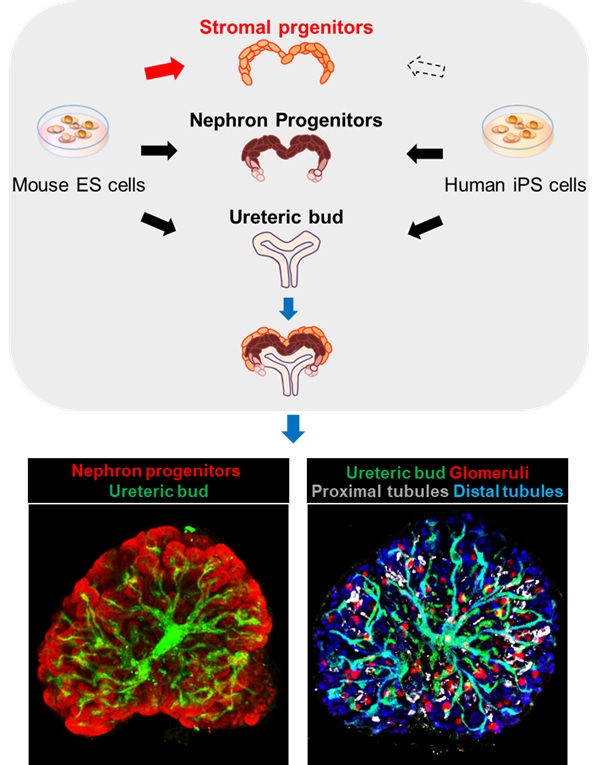
Figure
We developed a protocol to induce stromal progenitors from mouse ES cells (red arrow) and combined them with nephron progenitors and a ureteric bud, which were also derived from ES cells (black and blue arrows), to produce kidney tissue with a higher-order structure in which nephron progenitors and nephrons (glomeruli and renal tubules) are arranged around branching ureteric buds. We have already reported the methods to induce nephron progenitors and ureteric buds from human iPS cells, and the development of a stromal progenitor induction protocol from human iPS cells is awaited (dotted arrow).