
Akinobu Onitake, Yuka Matsushita-Ishiodori, Ai Johjima, Masatoshi Esaki, Teru Ogura, and Kunitoshi Yamanaka , The C-terminal α -helix of SPAS-1, aCaenorhabditis elegans spastin homologue, is crucial for microtubule severing. Journal of Structural Biology, in press
Spastin belongs to the meiotic subfamily, together with Vps4/SKD1, fidgetin and katanin, of AAA (ATPases associated with diverse cellular activities) proteins, and functions in microtubule severing. Interestingly, all members of this subgroup specifically contain an additional α -helix at the very C-terminal end. To understand the function of the C-terminal α -helix, we characterised its deletion mutants of SPAS-1, a Caenorhabditis elegansspastin homologue, in vitro and in vivo . We found that the C-terminal α -helix plays essential roles in ATP binding, ATP hydrolysing and microtubule severing activities. It is likely that the C-terminal α -helix is required for cellular functions of members of meiotic subgroup of AAA proteins, since the C-terminal α -helix of Vps4 is also important for assembly, ATPase activity and in vivo function mediated by ESCRT-III complexes.
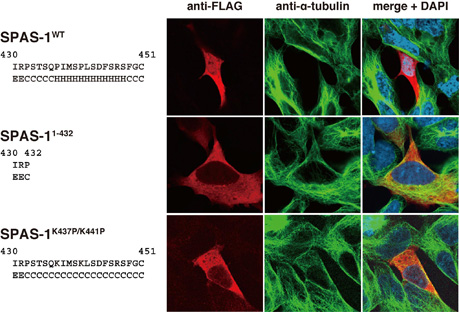
Figure: Requirement of the C-terminal α -helix of SPAS-1 for microtubule severing
When FLAG-tagged SPAS-1 WT was expressed in HEK293 cells, microtubule network disappeared. However, when C-terminal α -helix mutants (FLAG-SPAS-1 1-432 and FLAG- SPAS-1 K432P/K441P ) were expressed, microtubule network remained. These results clearly demonstrate that the C-terminal α -helix of SPAS-1 plays an essential role in microtubule severing.