
A synthetic nanofibrillar matrix promotes in vitro hepatic differentiation of embryonic stem cells and induced pluripotent stem cells
J Cell Sci 2013 126(23): 5391-5399
Taiji Yamazoe, Nobuaki Shiraki, Masashi Toyoda, Nobutaka Kiyokawa, Hajime Okita, Yoshitaka Miyagawa, Hidenori Akutsu, Akihiro Umezawa, Yutaka Sasaki, Kazuhiko Kume, and Shoen Kume
Lines of evidence have demonstrated that embryonic stem (ES) cells recapitulate normal developmental processes, and suggested that ES cells provide an attractive source for routine access to a large number of cells for investigation and therapies. We previously reported that ES cells cultured on a mesonephric cell line, M15 cells, or a synthesized basement membrane (sBM) substratum, with the addition of specific growth factors efficiently differentiated into an endodermal fate and subsequently adopted fates of various digestive organs, such as the pancreas, intestine and liver (Shiraki et al ., Stem Cells, 2008; Shiraki et al ., Genes Cells, 2008; Umedaet al ., Stem Cell Res, 2013; Ogaki et al ., Stem Cells, 2013; Higuchi et al ., J Cell Sci, 2010; Shiraki et al ., PLoS ONE , 2011 ) .
Here, we established a novel hepatic differentiation procedure using the synthetic nanofiber (sNF) as a cell culture scaffold. We first compared endoderm induction and hepatic differentiation between murine ES cells grown on sNF and several other substrata. F unctional assays for hepatocytes reveal that ES cells grown on sNF were directed into hepatic differentiation. Moreover, we established a completely chemical defined method, which requires no serum or no xenogenic substrata, thereby eliminating the risk of contamination with unknown factors. This method therefore enables a further investigation for stem cell biology and stem cell therapy. When human ES cells or induced pluripotent stem cells were cultured on nanofiber, Similar results were obtained.
Next, to clarify the mechanisms for the promotion of ES cell differentiation in the sNF system, we focused on the function of Rac1, which is a Rho family member protein known to regulate the actin cytoskeleton. There is evidence that cells cultured on nanofiber exhibit an in vivo like morphology (spheroid and dome formation), through associating with cytoskeletons. It is reported that murine ES cell can self-renew more rapidly on nanofiber plates than 2D plates. The proliferation was accelerated via Rac1 activation. W e found that activation of Rac1 occurred in undifferentiated and differentiated ES cells cultured on sNF plates, but not in those cultured on normal plastic plates. We also show ed that inhibition of Rac1 blocked the potentiating effects of sNF on endoderm and hepatic differentiation throughout the whole differentiation stages. Taken together, our results suggest that morphological changes result in cellular differentiation are controlled by Rac1 activation, and that motility is not only the consequence, but is also able to trigger differentiation. In conclusion, we showed that sNF is a promising material that might contribute to tissue engineering and drug delivery.
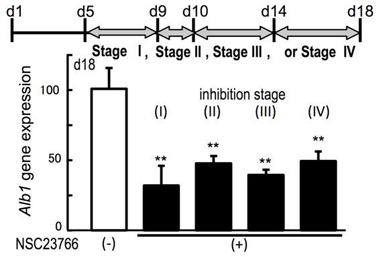
Fig. Inhibition of the Rac1 pathway blocks the differentiation-potentiating activity of sNF.
The expression of Alb1 on day 18 in differentiated cells, treated with (+) or without (-) the Rac1 inhibitor NSC23766 (100 mM) at indicated stages. Data shown represent mean ± s.e.m. (n=3); *P<0.05, and **P<0.01, compared with untreated cells on sNF with the Rac1 inhibitor by one-way ANOVA with the post-hoc Dunnett’s test.